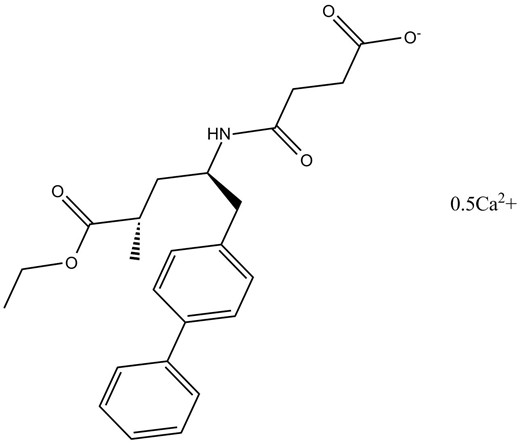
Sacubitril Hemicalcium
| 沙库比曲半钙盐 | Sacubitril Hemicalcium | 1369773-39-6 | In-House |
| 沙库比曲钠盐 | 149690-05-1 | In-House | |
| LCZ-4 | 1426129-50-1 | In-House | |
| LCZ-7 | 1012341-48-8 | In-House | |
| LCZ-8 | 1012341-50-2 | In-House | |
| LCZ-9 | 149690-12-0 | In-House |
Background
AHU-377 hemicalcium salt is a hemicalcium salt form of AHU-377. It is an inhibitor of neprilysin with IC50 value of 5 nM [1].
AHU-377 and the angiotensin II AT1 receptor antagonist valsartan compose LCZ696 in a 1:1 molar ratio. LCZ696 is an angiotensin receptor neprilysin inhibitor. It can reduce blood pressure and may be a novel drug for the treatment of heart failure. AHU-377 is a pro-drug, it can be converted by enzymatic cleavage of the ethyl ester into the active form LBQ657. It is reported that AHU-377(30 and 100 mg/kg, PO) can cause antihypertensive effect in a dose-dependent manner in DAHI-SS rats. But in the DOCA-salt hypertensive rats, it shows a weak reduction [2, 3].
References:
[1] Ksander GM, Ghai RD, deJesus R, Diefenbacher CG, Yuan A, Berry C, Sakane Y, Trapani A. Dicarboxylic acid dipeptide neutral endopeptidase inhibitors. J Med Chem. 1995 May 12; 38(10):1689-700.
[2] Voors AA, Dorhout B, van der Meer P. The potential role of valsartan + AHU377 ( LCZ696 ) in the treatment of heart failure. Expert Opin Investig Drugs. 2013 Aug;22(8):1041-7.
[3] Laxminarayan G Hegde, Cecile Yu, Cheruvu Madhavi et al. Comparative efficacy of AHU-377, a potent neprilysin inhibitor, in two rat models of volume-dependent hypertension. BMC Pharmacology 2011, 11(Suppl 1):P33.
Chemical structure





Proposal 18 Quality Consistency Evaluation projects which have approved 4, and 6 projects are under approving.

Advanced international quality management system has laid solid foundation for sales.

Quality supervision runs through the whole life cycle of the product to ensure the quality and therapeutic effect.

Professional Regulatory Affairs team supports the quality demands during the application and registration.


Korea Countec Bottled Packaging Line


Taiwan CVC Bottled Packaging Line


Italy CAM Board Packaging Line

German Fette Compacting Machine

Japan Viswill Tablet Detector

DCS Control Room










